In operation since 2006, Engemed Indústria results from years of dedication and investment in Research and Development, as well as the constant teaching and training of its highly specialized technicians and engineers.
100% Brazilian, our services constitute an opportunity especially for foreign manufacturers who wish to make their equipment viable in Brazil. We are certified in good manufacturing practices by Anvisa and strictly follow Brazilian regulatory demands.
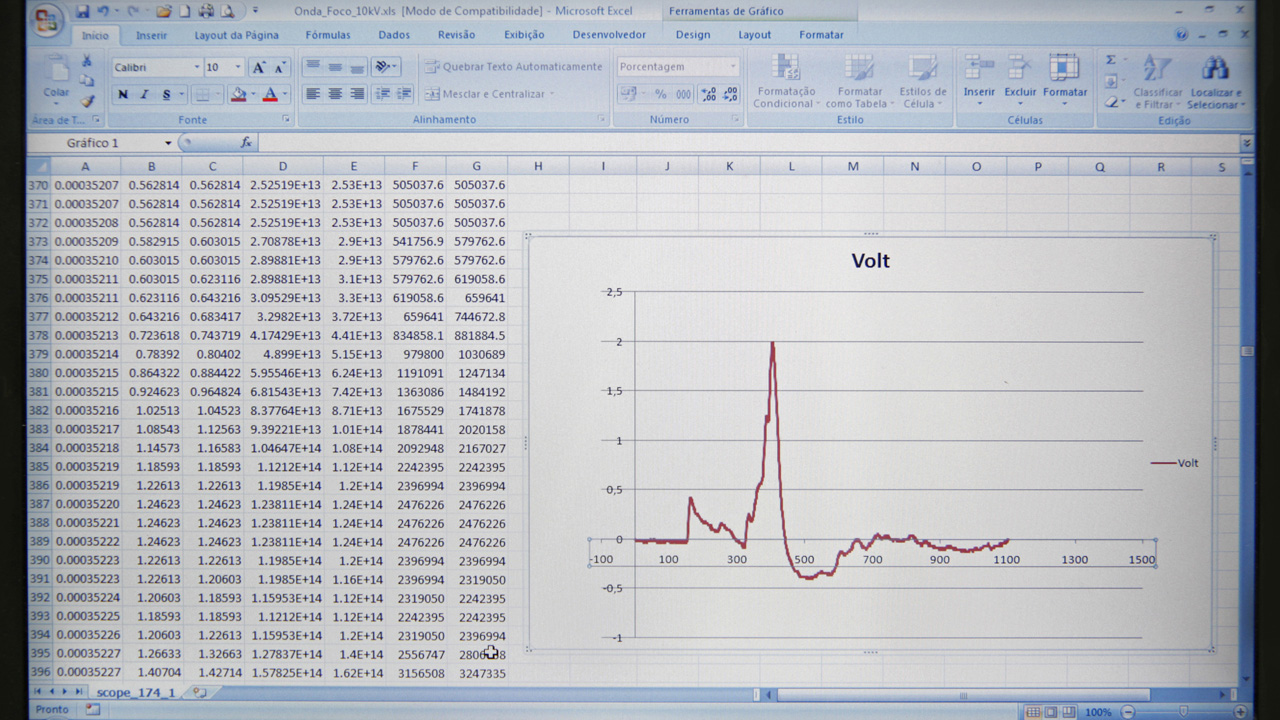
All medical devices are produced by qualified professionals, following internal standard procedures and the technical directives applied to each piece of equipment. During the assembly process, tests are run in order to ensure products have been made within the established standards.
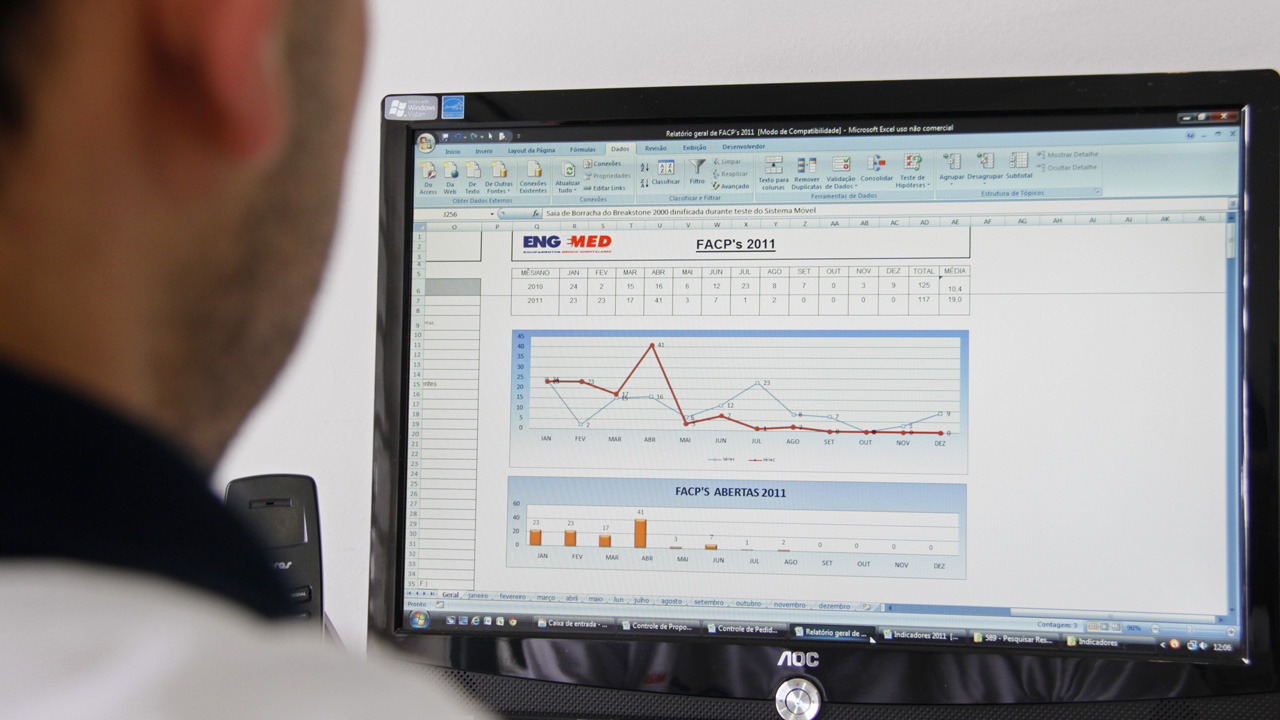
To ensure high quality products, Engemed keeps a Quality Control Department that inspects and carries out final tests in all produced devices so that they can be approved and okayed for purchase by clients. Every product is tested to make sure they comply with the directives and technical standards followed by the company.
Engemed has a warehouse which was especially built to store finished products and prevent them from physical damage and contamination before sales.
In order to guarantee maximum availability and promptness in helping clients, Engemed keeps a large stock of parts and pieces. We make use of a constant replenishment policy, receipt and storage procedures and periodical stocktaking to ascertain the number and integrity of stored materials. All materials are labeled accordingly so that both obsolete and damaged items can be removed from the warehouse.